CHEMetrics, Water Quality
-
WARNING! This product can expose you to chemicals including ethylene glycol, which is known to the State of California to cause birth defects or other reproductive harm. For more information go to www.P65Warnings.ca.gov .
-
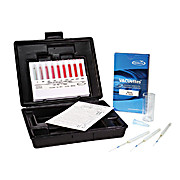
Hardness is a measure of the mineral content of water. Calcium and magnesium are the most common minerals that contribute to hardness. Hard water causes scaling in boilers and other industrial equipment, and diminishes the effectiveness of soaps and detergents. The EGTA Method (Calcium) …
-
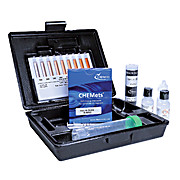
 Sulfide CHEMets® Kits
CHEMetrics
Sulfide CHEMets® Kits
CHEMetricsSulfides are naturally present in ground waters as a result of leaching from sulfur-containing mineral deposits. Surface waters do not usually contain high sulfide concentrations. Sulfides result from the decomposition of organic matter, from bacterial sulfate reduction under anaerobic conditions…
-
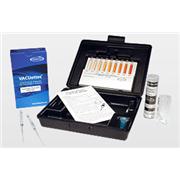
Glycol CHEMets Kit
CHEMetricsEthylene glycol and propylene glycol are the primary ingredients in commercially-available antifreezes. They are used with various corrosion inhibitors to protect metal surfaces in cooling water systems. CHEMetrics glycol kits are used to monitor potable waters for glycol contamination…
-
 Chlorine (Free & Total) CHEMets® Kits
CHEMetrics
Chlorine (Free & Total) CHEMets® Kits
CHEMetricsBecause of its strong oxidizing properties, chlorine is an excellent biocide used to treat potable waters, municipal wastes, and swimming pools. When used to treat potable water, chlorine helps alleviate the adverse effects of iron, manganese, ammonia, and sulfide. The Maximum Residual Disinfectant…
-
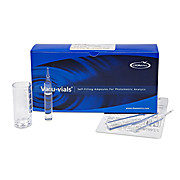
Copper (Soluble) Vacu-vials Kit
CHEMetricsCopper is naturally present in the earth’s crust and in seawater. Copper-containing fungicides are used to control biological growth in water supplies. The Maximum Contaminant Level Goal for copper is 1.3 mg/L in drinking water. The measurement of copper is an important means of monitoring…
-
Iron is present in nature in the form of its oxides, or in combination with silicon or sulfur. The soluble iron content of surface waters rarely exceeds 1 mg/L, while ground waters often contain higher concentrations. The National Secondary Drinking Water Standard for iron is 0.3 mg/L, as iron…
-
Method Dissolved carbon dioxide (CO2) is naturally present as a result of animal respiration, the decay of organic matter, and the decomposition of certain minerals. It is the major source of acidity in unpolluted water samples. Surface waters typically contain less than 10 ppm…
-
Bromine CHEMets Kit
CHEMetricsBromine, a less volatile compound than chlorine, is used as a sanitizing agent in drinking water systems, swimming pools, and spas. The bromine test method employs the DPD chemistry. Potassium iodide is added to the sample before analysis. Bromine reacts with the iodide to liberate iodine. The…
-
Chromate (Hexavalent) CHEMets Kit
CHEMetricsHexavalent chromium salts are used in numerous industrial processes. They are also used extensively as corrosion inhibitors in open and closed cooling water systems. The Diphenylcarbazide Method References: APHA Standard Methods, 22nd ed., Method 3500-Cr B - 2009. ASTM D 1687-02,…
-
Nitrite Vacu-vials Kit
CHEMetricsNitrite, an intermediate in the nitrogen cycle, is formed during the decomposition of organic matter but readily oxidizes to form nitrate. These processes occur in wastewater treatment plants, water distribution systems, and natural waters. Nitrites are useful as corrosion inhibitors,…
-
Hydrogen peroxide is a strong oxidizing agent with a variety of uses. Applications include the treating of industrial effluents and domestic waste and serving as a disinfectant in aseptic packaging. The Ferric Thiocyanate Method Reference: D. F. Boltz and J. A. Howell, eds.,…
-
Sulfite (free) Titrets® Titration Cells
CHEMetricsSulfite is not usually present in surface waters. If sulfite is discharged in effluents or from domestic wastewaters, it readily oxidizes to form sulfate. Sodium sulfite is the most common form of sulfite and is an excellent reducing agent with applications as an oxygen scavenger. Sulfite…
-
Ammonia Vacu-vials Kit, Range: 0-14.0 ppm
CHEMetricsLow-level ammonia nitrogen may be naturally present in water as a result of the biological decay of plant and animal matter. Higher concentrations in surface waters can indicate contamination from waste treatment facilities, raw sewage, industrial effluents (particularly from petroleum refineries),…
-
Nitrite VACUettes Kit, Range: 0-80 ppm
CHEMetricsNitrite, an intermediate in the nitrogen cycle, is formed during the decomposition of organic matter but readily oxidizes to form nitrate. These processes occur in wastewater treatment plants, water distribution systems, and natural waters. Nitrites are useful as corrosion inhibitors,…
-
Ammonia Vacu-vials Kit, Range: 0-7.00 ppm
CHEMetricsLow-level ammonia nitrogen may be naturally present in water as a result of the biological decay of plant and animal matter. Higher concentrations in surface waters can indicate contamination from waste treatment facilities, raw sewage, industrial effluents (particularly from petroleum refineries),…
-
Iron is present in nature in the form of its oxides, or in combination with silicon or sulfur. The soluble iron content of surface waters rarely exceeds 1 mg/L, while ground waters often contain higher concentrations. The National Secondary Drinking Water Standard for iron is 0.3 mg/L, as iron…
-
Phosphate CHEMets® Kits
CHEMetricsPhosphorus occurs naturally in rock formations in the earth's crust, usually as phosphate. High phosphate concentrations in surface waters may indicate fertilizer runoff, domestic waste discharge, or the presence of industrial effluents or detergents. Although phosphates from these sources are…
-
Iron in Brine CHEMets Kit
CHEMetricsIron is present in nature in the form of its oxides, or in combination with silicon or sulfur. The soluble iron content of surface waters rarely exceeds 1 mg/L, while ground waters often contain higher concentrations. The National Secondary Drinking Water Standard for iron is 0.3 mg/L, as iron…
-
Water Purification System Accessories
CHEMetricsThe CHEMetrics accessory is suitable for use with water purification system, which ensures best water quality by resisting airborne contaminants. This accessory is carefully designed with durable construction to ensure optimum performance.
-
Because of its strong oxidizing properties, chlorine is an excellent biocide used to treat potable waters, municipal wastes, and swimming pools. When used to treat potable water, chlorine helps alleviate the adverse effects of iron, manganese, ammonia, and sulfide. The Maximum Residual Disinfectant…
-
Kit comes in a plastic case and contains everything needed to perform 30 tests (except distilled water): Refill, Low and High Range Comparators, dilutor snapper cup, micro test tube, and instructions. Range: 0-1000 & 1000-10,000 ppm MDL: 100 ppm Method: Direct Nesslerization …
-
Hydrazine CHEMets Kit
CHEMetricsHydrazine is a powerful reducing agent that is used in various chemical processes and in boiler water as an oxygen scavenger. To control corrosion, residual hydrazine typically is maintained in the 0.05 to 0.1 mg/L range. Higher levels may be used to guard against corrosion when the boiler is out…
-
 Dissolved Oxygen CHEMets® Kits
CHEMetrics
Dissolved Oxygen CHEMets® Kits
CHEMetricsThe level of dissolved oxygen in natural waters is often a direct indication of quality, since aquatic plants produce oxygen, while microorganisms generally consume it as they feed on pollutants. At low temperatures the solubility of oxygen is increased; during summer, saturation levels can be as…
-
Ozone SAM Kit
CHEMetricsOzone is a strong oxidizing agent and is used as an alternative to chlorine as a biocide in the disinfection of drinking water. Ozone is used to remove odor, decolorize, and to control algae and other aquatic growths. Ozone is also used in various disinfectant and sterilization processes in…
-
Ammonia Accessory Solutions Pack
CHEMetricsQty. per pack: Two 10mL bottles of A-1404 Stabilizer Solution Two 10mL bottles of A-1405 Catalyzer Solution Two 10mL bottles of A-1406 Activator Solution Shelf-life: 8 months The A-1410 Accessory Solutions Pack supplies enough of each solution to perform approximately 80 tests.
-
Hydrogen peroxide is a strong oxidizing agent with a variety of uses. Applications include the treating of industrial effluents and domestic waste and serving as a disinfectant in aseptic packaging. For the food and beverage industry, CHEMetrics Hydrogen Peroxide CHEMets® and…
-
Hydrogen peroxide is a strong oxidizing agent with a variety of uses. Applications include the treating of industrial effluents and domestic waste and serving as a disinfectant in aseptic packaging. The Ferric Thiocyanate Method Reference: D. F. Boltz and J. A. Howell, eds.,…
-
Kit comes in a plastic case and contains everything needed to perform 30 tests (except distilled water): Refill, Low and High Range Comparators, dilutor snapper cup, micro test tube, and instructions. Range: 0-30 & 30-300 ppm MDL: 5 ppm Method: Direct Nesslerization …
-
Iron is present in nature in the form of its oxides, or in combination with silicon or sulfur. The soluble iron content of surface waters rarely exceeds 1 mg/L, while ground waters often contain higher concentrations. The National Secondary Drinking Water Standard for iron is 0.3 mg/L, as iron…
-
Sulfide Vacu-vials® Kits
CHEMetricsSulfides are naturally present in ground waters as a result of leaching from sulfur-containing mineral deposits. Surface waters do not usually contain high sulfide concentrations. Sulfides result from the decomposition of organic matter, from bacterial sulfate reduction under anaerobic conditions…
-
Nitrate Vacu-vials Kit, Range: 0-50.0 ppm
CHEMetricsNitrate is the most completely oxidized form of nitrogen. It is formed during the final stages of biological decomposition, either in wastewater treatment facilities or in natural water supplies. Low-level nitrate concentrations may be present in natural waters. However, a Maximum Contaminant Level…