
Specifying a high grade versus a standard grade can be more cost-effective in the end for some applications because reduced rework and material consumption will justify the higher initial cost.

The use of more specialized and rigorously tested acetonitrile grades is becoming more common, as laboratories seek to remove the uncertainty of solvent performance from their work.
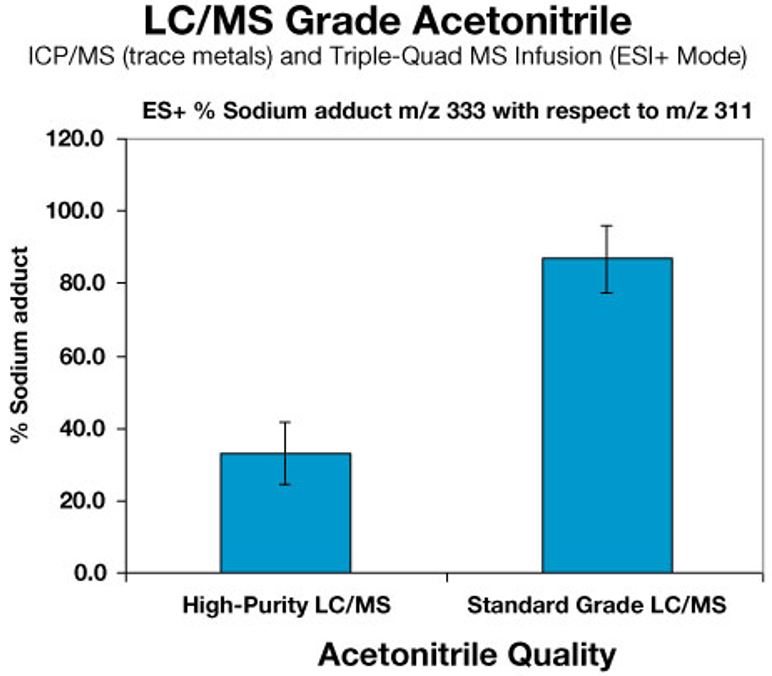
(Figure 1) Ion suppression (e.g., sodium adduct formation) can be a common issue in advanced LC/MS applications, often causing misinterpretation of data. Functional-use testing and testing for trace metals in the parts-per-billion range ensures that contaminants which may produce unclear or flawed results are not present.
| Acetonitrile is one of the most frequently used chromatography solvents, available in such a wide range of quality (grades) that selecting the most appropriate grade can be a critical and challenging decision. Grade selection can be based on key factors that include the degree of precision required, the specific application involved and the scale of chromatography. As laboratories seek to remove the uncertainty of solvent performance from their work, the use of a more specialized and rigorously tested grade is becoming more common. For example, exacting trace analysis in complex matrices using high-performance liquid chromatography (HPLC) or liquid chromatography/mass spectrometry (LC/MS) requires a specialized grade of acetonitrile to meet stringent performance specifications. Laboratories can realize these five benefits when selecting the most appropriate acetonitrile grade: - Minimal rework
- Greater reproducibility of results
- Less challenging methods transfer
- Improved instrument sensitivity to both small and large molecule detection in drug discovery
- Greater accuracy – including fewer erroneous peaks that are caused by metal adduct formation, as well as extended column life
Such benefits can offset any additional cost for the higher-purity product, making the selection of a better grade a worthwhile purchase. In function-tested LC/MS applications where the minimization of contaminants and trace metals is critical to ion formation or related reactions within the sample (Figure 1), increased sensitivity and reliable results alone can justify a higher initial material cost. Alternatively, routine, high-volume and well-characterized applications like pharmaceutical or food and beverage quality control may use a standard grade for balancing budgetary goals while delivering acceptable results. For these applications, it is not cost-effective to over-specify for a high-purity grade where it is not needed. Selecting the appropriate acetonitrile grade for HPLC and LC/MS applications can be broken down into whether high or standard grades are needed. The following table provides some key specifications for comparison of standard- and high-grade HPLC and LC/MS acetonitrile. |